Oxidic decorations induce a surface charge which depends on their acidity (acidic oxides lead to a negative surface charge), affecting surface defect concentrations, any existing surface potential step, potentially adsorption dynamics, and consequently also the OER kinetics.ĭefect chemistry mixed ionic electronic conductors oxygen exchange pulsed laser deposition surface decoration surface potential. 
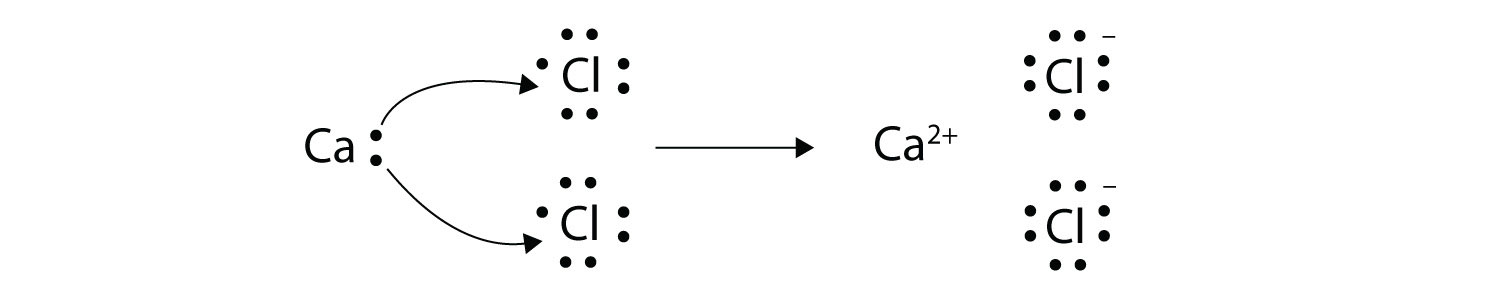
From a mechanistic point of view, our results indicate a correlation between the surface potential and the altered oxygen exchange activity. NAP-XPS was further employed to investigate changes of the surface potential step on decorated surfaces. This is confirmed by NAP-XPS measurements which find only minor changes of the Pr-oxidation state upon decoration. Furthermore, the total conductivity of the thin films does not change upon decoration, indicating that defect concentration changes are limited to the surface layer. Now, sodium is located in group 1 of the Periodic Table, which means that an atom of sodium has Math Processing Error valence electron. While a significant alteration of the OER rate was observed after decoration with binary oxides, the pO 2 dependence of the surface exchange resistance and its activation energy were not affected, suggesting that surface decorations do not alter the fundamental OER mechanism. Explanation: The first thing to notice here is that this compound contains a metal, sodium, and a nonmetal, oxygen, so right from the start, you know that youre dealing with an ionic compound. The surface chemistry of the electrodes was investigated by near-ambient pressure XPS measurements (NAP-XPS) at elevated temperatures and by low-energy ion scattering (LEIS). The oxygen exchange reaction (OER) rate and the total conductivity were measured by in situ PLD impedance spectroscopy ( i-PLD), which allows to directly track changes of electrochemical properties after each deposited pulse of surface decoration. The oxygen exchange kinetics of epitaxial Pr 0.1Ce 0.9O 2-δ electrodes was modified by decoration with submonolayer amounts of different basic (SrO, CaO) and acidic (SnO 2, TiO 2) binary oxides.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
